Pharma Tablet Press Technology Transfer & Process Optimisation for Ibuprofen based Formulation
Laphal Industries purchased Prexima 300, a middle-size industrial tablet press from Ima and explored the reliability and flexibility of the machine. The company was also able to find out the best process parameters according to its requirements.
Related Vendors

In the pharmaceutical industry’s competitive and ever-changing business climate, companies are frequently divesting certain assets and acquiring others. As a result, the industry is often very busy with manufacturing site changes and drug product technology transfers.
According to ICH10, the guideline for pharmaceutical quality system “The goal of technology transfer activities is to transfer product and process knowledge between development and manufacturing, and within or between manufacturing sites to achieve product realisation. This knowledge forms the basis for the manufacturing process, control strategy, process validation approach and ongoing continual improvement.”
It is necessary to start from a definition: Receiving Unit (RU) is the involved disciplines at an organisation where a designated product, process or method is expected to be transferred, whereas the Sending Unit (SU) represents the involved disciplines at an organisation from where a designated product, process or method is expected to be transferred.
During the transfer process, the RU should identify any differences in facilities, systems and capabilities and communicate with the SU about these differences to understand the potential impact on the ability to run the process to deliver good product quality. Differences should be understood and satisfactorily addressed to assure equivalent product quality. Based on the information received from the SU, the RU should consider its own capability to manufacture and pack the product to the required standards and should develop relevant plant operating procedures and documentation before the start of production.
Buying a new machine involves major change control as well as technology transfer activities if the production is actually ongoing at the customer’s site. Verifying the impact on the ability to run the process is synonymous of performance qualification. It represents a documented verification that the equipment or system operates consistently and gives reproducibility within defined specifications and parameters for prolonged periods. The process validation, necessary in this case, actually provides a high degree of assurance that a specific process will consistently result in a product that meets its predetermined specifications and quality characteristics.
The aim of this study is technology transfer and process optimisation in the context of a cooperation between Ima (Italy) & Laphal Industries (France): in particular, Laphal bought a middle-size industrial tablet press from Ima. In the first part of the study, placebo from the customer was tested to explore the reliability and flexibility of the Ima machine as well as to find the best process parameters according to Laphal’s needs. The parameters found was secondly reproduced and adapted with the real ibuprofen-based formulation to confirm the equivalency in terms of product quality.
Materials & Methods
To analyse the reliability and capacities of the machine, a placebo formulation, containing lactose, cellulose and magnesium stearate, was used.
The trials were conducted by using the middle size equipment, Prexima 300 (Ima, Italy) hosting 33 station Euro-B turret and round-shaped bi-convex punches (diameter 8 mm).
In order to qualify the equipment, different tests were performed to study:
1. The influence of pre-compression force (PC or PCF): At five different tablet press speed (53, 76, 92, 98 and 105 rpm) different pre-compression forces were tested (0, 2, 4, 6, 8,4 kN). To evaluate which is the best combination, tablet strength and its relative deviation were explored.
2. The influence of feeder speed: According to the results obtained for test 1, different paddle speed was performed (12, 30, 50, 70, 90, 120 and 140 rpm). To estimate the best choice, relative standard deviation (RSD %) even on weight than on hardness was considered: the lower the better.
3. The influence of compression force (CF): Maintaining the PCF half of the CF studied, trials were executed at the same tablet press speed. At the end, tablet strength and its RSD (%) were the results that were considered.
4. The influence on strength with different pre-compression and compression forces: The aim of the test was to define a better ratio between PCF and CF while maintaining the same tablet press speed, paddles speed and dosage chamber (that means same tablet weight).
The tests that were performed can be summarised in the following diagram:
Once again, tablet strength and its relative standard deviation were taken in consideration to find the best process parameters.
5. Hysteresis study: The study focused on weight hysteresis (%) when the tablet press parameters were changed according to test 4 (PCF and CF). First, the average weight of 20 tablets was evaluated. Secondly, at a fixed compression force and tablet press speed, weight variation was analysed for 20 tablets while changing the PCF (0, 2, 4, 6, 8,4 kN). The acceptable hysteresis limit was among 1,4 % and 1,9 %.
Once the best conditions were defined, machine yield and continuous production of 200 kg was executed. Starting from the knowledge acquired with the placebo formulation, the customer’s ibuprofen blend was tested in Prexima.
Results & Discussion
The tablets were always produced according to the target expected on weight, thickness and hardness: the machine’s performance convinced the customer to buy the machine for his purposes.
1. Influence of pre-compression force
Maintaining the same weight, tablet strength and its RSD (%) can be summarised in a graph (Figure 1, Figure 2)
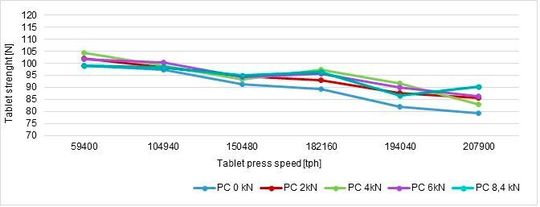
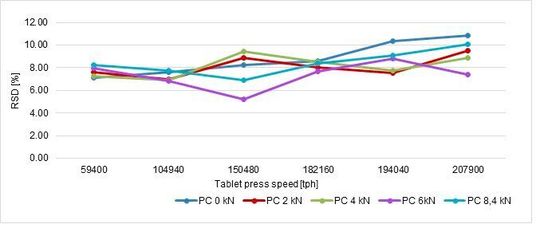
In conclusion, the best parameters are obtained at 93 rpm (182160 tph) without particular influence of the pre-compression force even if this is a critical tableting process parameters as reported in the previous article.
2. Influence of feeder speed
Maintaining the same weight, thickness and hardness. The paddle speed was changed to find the highest possible stability: this reliability was analysed by monitoring RSD (%) on weight and hardness (Figure 4, Figure 5).
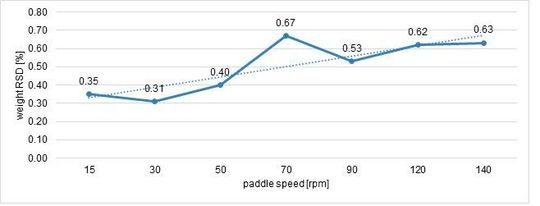
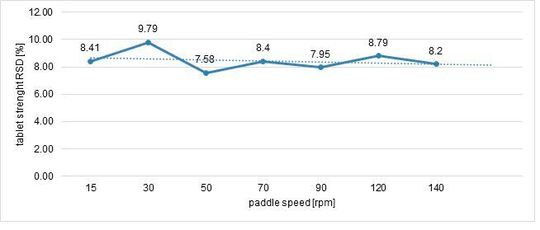
The highest stability was reached with 30 rpm as paddle speed.
3. Influence of compression force
Following the same approach, the tablet strength was analysed as an answer of compression force influence. Hardness RSD (%) is also important to understand process stability (Figure 6, Figure 7).
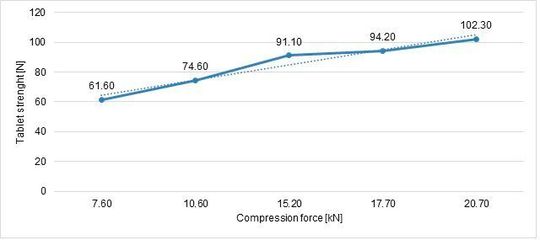
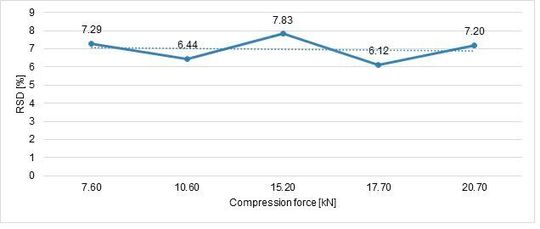
The tablet strength increases linearly with an increase of compression force, the best RSD was achieved at 17,7 kN.
4. Influence on strength with different pre-compression and compression forces
At the same tablet press speed, paddle speed and tablet weight, tablet strength and RSD were analysed and summarised in the following graphs (Figure 8, Figure 9).
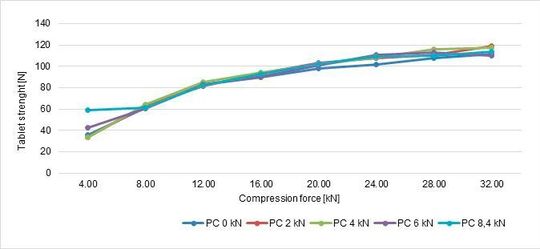
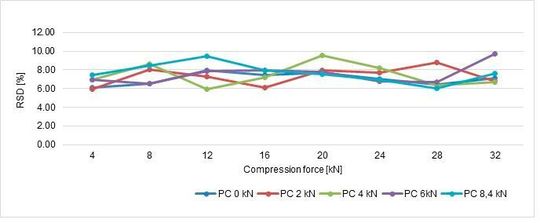
In conclusion, performing with different compression forces, the tablet strenght tends to increase until 28 kN, then there is a plateau meaning that the best values are achieved: if the compression force is further increased, probably lamination tends to appear. In terms of pre-compression, the best values are 6 and 8,4 kN.
5. Hysteresis study
First, weight hysteresis was monitored with parameters of test 4 considering as acceptable value 1,4 % and maximum 1,9 % (Table 1).
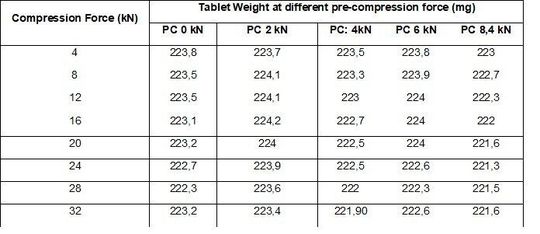
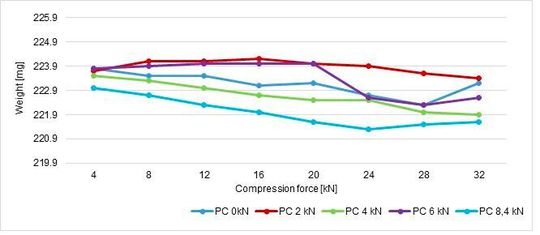
According to these values, the weight average is 223,02 mg and hysteresis of 1,4 % corresponds to 219,9 mg and 226,14 mg. If these limits are reported in a graph, the weight is in compliance with the hysteresis requested (Figure 10).
To confirm preliminary results, the weight variation was studied at 28 kN (tablet speed: 53 rpm) and different compression forces (Table 2).
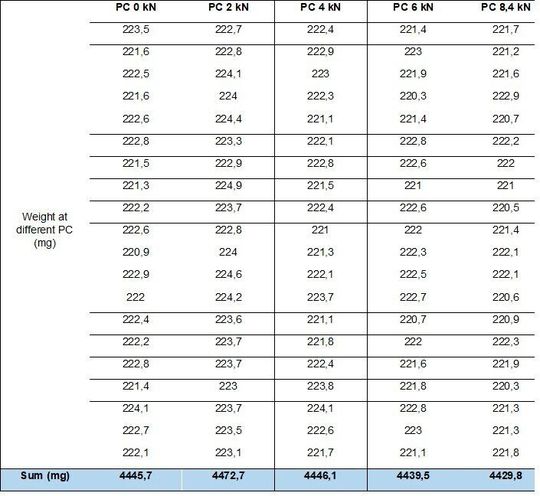
The average calculated on each test and on 20 tablets is 4446,76 mg with a hysteresis of 1,4 %, limits acceptable are between 4509 mg and 4384,5 mg: all values are within limit and real hysteresis is 0,3 %.
The performance test was executed to verify the capability of the machine: the machine performed at 92 rpm (182160 tph), 6 kN as PC and 21,5 kN as CF. 10,3 kg of powder was loaded in the machine and was made to run for 15 minutes continuously: after 14’54’’, the machine stopped and the tablets were unloaded – 10,127 kg was good and 0,00739 kg was rejected (total: 10,2 kg). Hence, the performance of the machine was 99 % meaning that almost all the powder which was loaded was compressed without significant losses.
The above performance was achieved without a significant temperature increase of the die plate. Thanks to the new-generation gearboxes implemented on Prexima, the temperature of the die plate was always <32°C without the need to apply specific and/or complex cooling systems which Laphal used to apply on other presses.
Once the tableting process was explored, production of 200 kg was performed at a turret speed of 76 rpm. The same tablet press speed as well as paddle speed was transferred for real ibuprofen production (table 3).
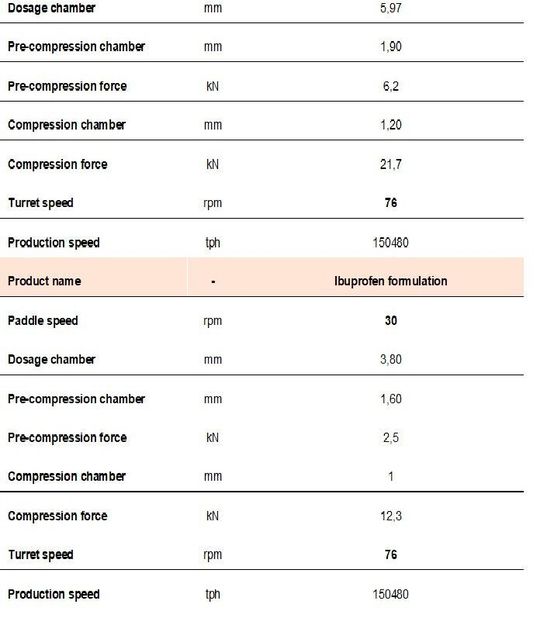
Because of the inner differences between placebo and the real API based formulation, PCF and CF were adapted.
Conclusions
The aim was achieved: preliminary trials with placebo allows the customer to understand the reliability and flexibility of Prexima 300. Secondly, process parameters were transferred successfully in real production, confirming the customer’s expectations and achieving the same level of quality obtained with the placebo formulation. The temperature of the die plate was also confirmed to be <32°C over the long-run production with Ibuprofen. Thanks to the cooperation between Ima and Laphal Industries, the technology transfer was quick and without any inconvenience.
(ID:47007863)



:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/7e/c87e276b243339dbff4a8eca8f67a55e/0130871216v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/ff/97ffc5d8894eaaabac89fa86c88b5c04/0130558883v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1a/64/1a64a67b53537b893f5a6509dee97d87/0131192946v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/72/f3/72f3a2fed9f27d1b33a66f3021eb6ed2/0131192934v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/35/74/357499c9bc77391babfd047b6d7d9e10/0131139412v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f1/87/f187c86e791dfcf8ecccf615a6a3d054/0131055400v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b4/e5/b4e5eecb07d3491bde7bf6a22602ab77/0131085753v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/a6/43a69c0b9e26996ab4f3971f202afd82/0131055378v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/7d/0e/7d0e9e3fa6a5289338833948b8c8e6d0/0130702623v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d6/c4/d6c445a05ff0384818acac8b3530e2d3/0130701377v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/79/7179c77f8bae9e48bf427708573ad6ac/0131057546v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3f/7f/3f7f7542cd5392be0f3074f074a75b36/0131055389v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/26/0e/260ee8cbbc76b6dcaa4d6221ecb44506/0131014543v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/94/0e/940e978ce75be13842063b9c81d5f66e/0130871936v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/0c/a1/0ca135e0486c9af90a0552aea85de88c/0130439525v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/113800/113818/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/2000/2093/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:quality(80)/p7i.vogel.de/wcms/9c/7b/9c7bfde0cb4213fc926662d1593718fc/0127804144v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/6f/276f0df09fd968e74c7c58d5915d07f1/0127144840v2.jpeg)