Patent Cliff How to manage the patent cliff?
Balancing between patent cliff, generic drugs and emerging markets the pharmaceutical industry has to position itself anew. This article is carrying out an analysis.
Related Vendors
Price Waterhouse Cooper (PWC) released the latest in a series of “Pharma 2020” reports in November 2012, called “Pharma 2020 — From Vision to Decision” states that Pharma’s future has never looked more promising — or more ominous. Major scientific, technological and socioeconomic changes will revive the industry’s fortunes in another decade, but capitalising on these trends will entail making crucial decisions first.
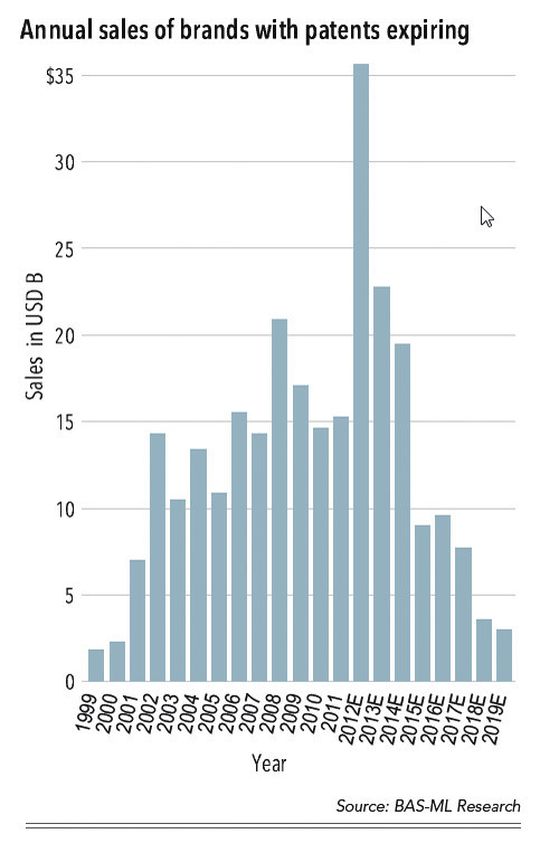
The statement hits the outlook well. 2012 was probably the toughest year of the pharmaceutical patent cliff and there seem to be two main agendas for pharmaceutical companies after the patent cliff: Either cut cost and streamline or invest and grow innovation. Both agendas will work but leads to very different implications for business and future investments.
For pharmaceutical companies in this rapidly changing world it will be important to select business partners and suppliers that understand their business. Because the business conditions change more rapidly than ever before, there is no time for selecting new partners. Therfore it is important to select the right partners and build long and trust-based relations that can ensure mutual business success.
Setting the Scene
The pharmaceutical industry is fundamentally healthy and has been so for many years. The global revenue in 2011 was estimated to $ 955 billion (whereas 1981 was approximately 70 billion) and although the growth rate is slightly decreasing. The overall market has been growing by approximately 9 % (compound annual revenue growth) in value over a period of 30 years and it is still expected to grow towards 2016.
Looking forward towards 2016 the main focus is on cancer and cancer-related diseases, diabetes and general antibiotics. Several new medicines are expected the coming years, including some that cover previously unmet medical needs in Alzheimer, autoimmune diseases, diabetes and a number of cancers and so-called orphan drugs. The top 20 therapy areas account for 42 % of the total drug spending.
As major patents expire, many companies face significant challenges to their future business and make decisions on investments, markets, site locations, products and technologies that are very different from before the patent challenge. For several years the number of new drug approvals has been decreasing, but the last couple of years we have seen a new increase in both Europe and the USA that increases the optimism.
Besides, the many products that have passed patent expiry, have a revival as leading generic drugs where they replace older and less effective medication at cost-effective prices. These new generics are encouraged by the increasing pressure from regulatory authorities and reimbursement policies towards the most cost-effective solutions and this pressure must be expected to continue in the future.
(ID:36102260)



:quality(80)/p7i.vogel.de/wcms/f1/41/f141fc30eddadc5a340d28fd8980aa27/0131438828v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/22/a4/22a4a5a34b3157c9ef8b6fdc5a2b11ec/0131429879v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cb/21/cb21c4c6df2f64cd4b6c321a94b8f89b/0131663349v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/56/62/5662647534f2d5e37b2611e648f0614a/0131662783v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/86/8486433528650e8b2a82a5632369d03b/0131635199v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/08/18/0818c09b149b4bf2c5e20ba853ccadba/0131577464v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/30/9c/309c19fe8590076dd4d7acd0e79fe962/0131651735v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/16/71167a6d12f3f694a9cf744a6f12e77b/0131584322v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f5/ea/f5eae046dd5abe4a43b65e4aee1d5784/0131429900v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/14/fc/14fcd8993b9bb6ad259d8598450cbd2f/0131662897v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/49/96/4996a6d6c3423e281010abb7392973e9/0131607502v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c4/8a/c48a75246f79f5e55f4f972301724e04/0131574854v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/12/59/125984e434f018f1c36129631804d8dd/0131469975v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/92/a892445199f1f77c914c4de44d10fa5d/0131584311v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2c/42/2c42affbdc6932f5a2fa8393b4fb3824/0131478702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/73/72/7372423f290265b4ab6020b9a9d8d804/0131663060v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ce/f0/cef0fd9512272577eb8432b4483b5d51/0131662913v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bd/ea/bdea404907a685e5265adc98538cce0d/0131662795v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ec/90/ec9035e68fff43666d2b5d94d2b9ba4d/0131633541v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/44/a5/44a5d4f0a4c4b001846500c9e853d821/0131634217v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/87/d087e325d2fc41b031fd812c7515d5f3/0131574190v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/109600/109698/65.png)
:fill(fff,0)/p7i.vogel.de/companies/5f/98/5f98fbb2e7bf2/05-trm-filter-logo-with-slogan-en-alt.png)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/64500/64561/65.jpg)
:quality(80)/p7i.vogel.de/wcms/74/af/74af2b65341eeee13a44bc85eb759596/0129839901v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8e/14/8e146e574e384cd7d3dabb404f8df971/0127923049v2.jpeg)