Computational Fluid Dynamics (CFD) CFD Expands Footprint into Pharmaceutical Production
Computational Fluid Dynamics (CFD) has a proven track record as a software simulation tool to support detail equipment design and development of entire systems. The pharmaceutical industry is now discovering the advantages of this approach.
Related Vendors
The Life Sciences are not new territory for CD-Adapco, according to Kristian Debus. However since the Sector Manager moved into his new role, the pace of activity has quickened and Debus, a good communicator who is originally from Munich and has worked in California for many years, now spends more of his time on the road. That simply comes with the new job. Although customers in the pharmaceutical and medical industries have been using the STAR-CCM+ simulation program for 15 years, the Life Science Sector Business Unit has only officially been in existence for about a year.
Debus has taken on the new challenge of absorbing and channeling momentum from the market and directing it into the development engineering departments. He also has responsibility for organizing internal training. Put simply, in a software company which has a workforce of 700, his job is to ensure that the pharmaceutical sector receives the emphasis which is commensurate with such a high-revenue industry.
“Continuous Pharmaceutical Production Has Considerable Potential”
Debus does not generally tick off a list of big names, but the fact that someone from Novartis held a key lecture at one of the CD-Adapco user conferences would indicate that the company is part of the customer base. Debus is convinced that continuous pharmaceutical production has considerable potential and should open more doors over the next few years.
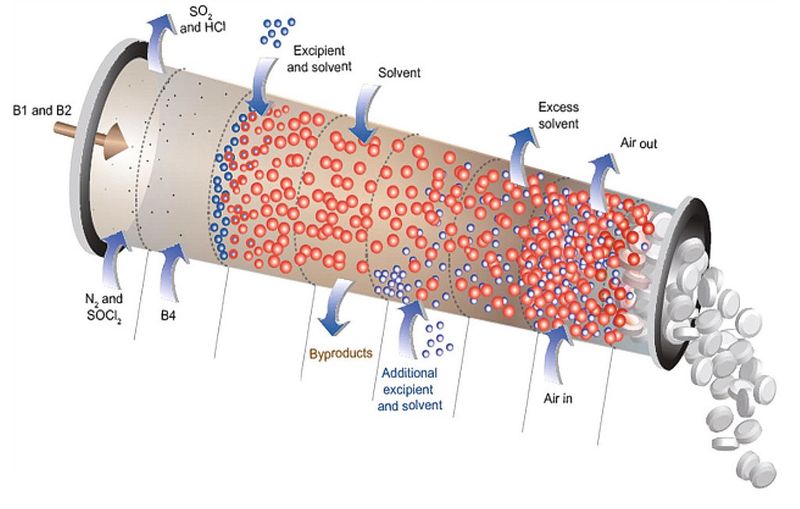
Since March, experts from CD-Adapco have been working as part of the C-SOPS (Center for Structured Organic Particulate Systems) consortium in conjunction with Prof. Ferdinando Muzzio from Rutgers University along with the University of Puerto Rico at Mayagüez, Purdue University, the New Jersey Institute of Technology and pharmaceutical companies which provide strategic direction (Pfizer, Glaxo Smith Kline, Johnson&Johnson, Merck, Lilly and Bristol Myers-Squibb) to develop a continuous process for API production which includes blending, granulation, tablet pressing and coating.
“The consortium is using STAR-CCM+’s DEM technology and analyzes the interaction of powders and particles in parallel with process development,” reported Debus. The combination of CFD and DEM (Discrete Element Modeling) makes it possible to analyze individual items such as tablets and capsules and understand how they interact with each other, e.g. in a fluidized bed or tablet coater. Every process step which the consortium develops is modeled and optimized in parallel, and appropriate sensor systems are deployed given the fact that PAT is so important in continuous production.
(ID:39595990)



:quality(80)/p7i.vogel.de/wcms/f1/41/f141fc30eddadc5a340d28fd8980aa27/0131438828v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/22/a4/22a4a5a34b3157c9ef8b6fdc5a2b11ec/0131429879v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/08/18/0818c09b149b4bf2c5e20ba853ccadba/0131577464v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4f/a5/4fa505865cd05830f66d98c7f0248d3c/0131576373v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/47/37/473728562889646fc39bffb62ec500ae/0131575786v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b2/f9/b2f9f1b88b4ee749df0ea3af3a7da90d/0131440719v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/16/71167a6d12f3f694a9cf744a6f12e77b/0131584322v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f5/ea/f5eae046dd5abe4a43b65e4aee1d5784/0131429900v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c4/8a/c48a75246f79f5e55f4f972301724e04/0131574854v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/12/59/125984e434f018f1c36129631804d8dd/0131469975v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/90/0990b4f93bc2629e6a76dd8d10c7ef9b/0131365319v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/18/42/1842c51b9071515986da6d691978da02/0131330223v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/92/a892445199f1f77c914c4de44d10fa5d/0131584311v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2c/42/2c42affbdc6932f5a2fa8393b4fb3824/0131478702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/42/4d/424d78ed7c10947d632e3415a6afc48d/0131583053v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/49/78/4978485187e68eff46feab3f9a85072f/0131578860v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/87/d087e325d2fc41b031fd812c7515d5f3/0131574190v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2e/a2/2ea266feec2d8d52270eaca7f41af6f7/0131478707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/113800/113818/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:quality(80)/p7i.vogel.de/wcms/01/15/0115415d906c21a45a3843617426db74/0125679758v3.jpeg)
:quality(80)/p7i.vogel.de/wcms/9c/7b/9c7bfde0cb4213fc926662d1593718fc/0127804144v1.jpeg)