Simultion for API Production Boost Your API Production with Numerical Simulation – From Batch to Continous Processing
Take your API production to the next level – Multi-physics numerical simulation is emerging as game-changing technology to help continuous manufacturing for Active Pharmaceutical Ingredients (APIs) become a reality through virtual prototyping, optimization and modeling of the complete system.
Related Vendors
The pharmaceutical industry is encountering a decline in productivity, and outdated “tried-and-true” batch processes are at the root of the problem. The batch-based systems currently in place are inefficient due to segmented steps involving multiple facilities and requiring start-and-stop of the batch, site-to-site transfer and warehouse storage.
Continuous manufacturing, a non-stop end-to-end manufacturing process, could modernize the industry and solve its productivity crisis. At a recent MIT conference, Josef Jimenez, CEO of Novartis, stated that changing production from batch to continuous will transform the way medicines are made around the world and could cut the time from development to market-entry in half. Implementation of these processes will result in smaller production plants, lower inventory costs, reduction in carbon footprint and higher quality products.
Simulation – A Reaction to Changing Regulatories?
The regulatory agencies are also starting to lay the groundwork for continuous manufacturing with several initiatives and regulatory frameworks such as the Process Analytical Technology (PAT) and Quality by Design (QbD). Each of these encourages the development of new manufacturing technologies by building quality into the process and using a science-based quantified risk approach.
Both the chemical and food processing industries have been improving their productivity by successfully integrating continuous manufacturing into their plants. It is clear that regulatory hurdles and conservative thinking by the pharmaceutical industry can no longer be used as an excuse to avoid taking pharmaceutical manufacturing into the 21st century.
How to Achieve Continious Pharmaceutical Production
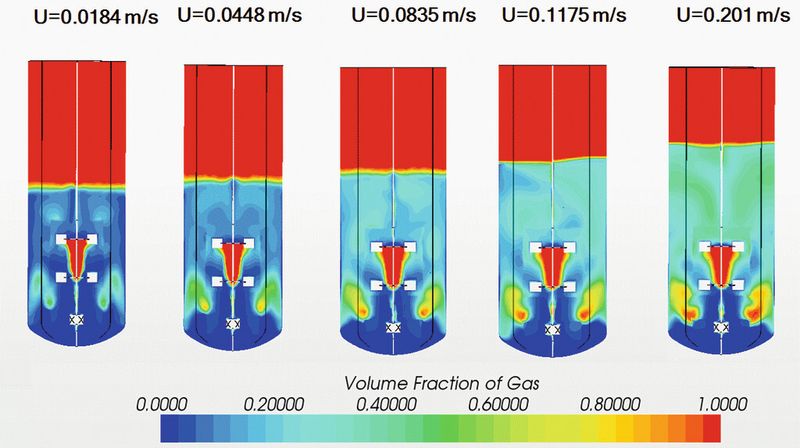
Before continuous manufacturing can become main-stream, potentially suitable candidate processes must be identified and designed, and risks must be analyzed and mitigated. This will help manage regulatory compliance and make a business case for implementation. Multi-physics Computational Fluid Dynamics (CFD), a numerical method for predicting the coupled behavior of fluid, gas and particulate flows including heat and mass transport, offers a solution for the enhanced understanding and design of these novel processes.
(ID:42311500)



:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/7e/c87e276b243339dbff4a8eca8f67a55e/0130871216v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/ff/97ffc5d8894eaaabac89fa86c88b5c04/0130558883v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/f4/54f4b4e2a86b203e0dfd84221602bdb5/0130367283v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/f6/98f605368ab5737a54c0e4489ad8cc3e/0131003694v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2e/c9/2ec993dc8f88d8f4ce78410d164c141e/0131003660v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/06/cf/06cfc293d1d767ba8aa5ef11bedb56c8/0131003115v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/9d/c1/9dc1459abca8c6eee52e304ae7125740/0131002528v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3b/7b/3b7bb6b7a7a8aa735ef9164789c529ea/0131023097v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/15/95/15952e5697fef87ba8da5a8f521dbfa5/0131006636v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bc/67/bc6743f143cc06e350a4a47e1e600399/0131002840v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e0/2e/e02e124a066eb32e81e6ad35e6b32681/0130798212v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/7d/0e/7d0e9e3fa6a5289338833948b8c8e6d0/0130702623v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d6/c4/d6c445a05ff0384818acac8b3530e2d3/0130701377v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/5d/575d8ae4e7f56748c0aa48ca34735b5d/0130646451v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/33/1c33cb645b2d07dacd829b45e08a932a/0130641492v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/98/989863664c7b65e87caf74a08239d487/0130167217v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/26/0e/260ee8cbbc76b6dcaa4d6221ecb44506/0131014543v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/94/0e/940e978ce75be13842063b9c81d5f66e/0130871936v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bb/d8/bbd8135709bd2fae7fe813bc268feea0/0130871485v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4b/d9/4bd9d4ef05d8bd7fdf78eca991ced8de/0130871466v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/0c/a1/0ca135e0486c9af90a0552aea85de88c/0130439525v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/e3/09e378194b496921d2a267a6752322eb/0130333473v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/2000/2093/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:quality(80)/p7i.vogel.de/wcms/41/85/41850761720b85d6be9d51332da2b515/0125589239v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/df/cd/dfcd0c2e0eb4b554f956e0161ae08519/0129794328v1.jpeg)