Catalysts Process Engineers Use New Catalysts to Solve Old Problems
Catalysts, whether heterogeneous or homogenous, ignite the turbo in reactors and have what it takes to make future dreams come true. The wish list includes things like the age-old dream of using carbon dioxide as a chemical feedstock.
Related Vendors
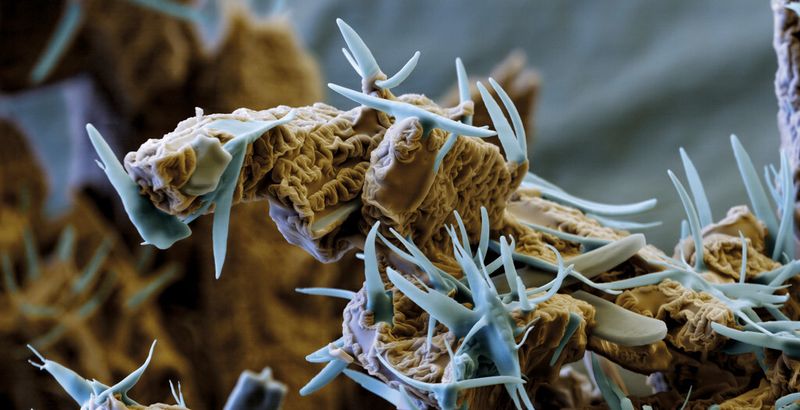
Haber-Bosch synthesis is the classic heterogeneous catalytic technique, and 99.9 percent of all chemists could probably recite the details of this first large-scale industrial process while still half asleep. Times have changed. Ammonia synthesis was a revolution in 1913, but today it is part of the undergraduate curriculum. Maybe that is one reason why people tend to forget that the underlying importance of reactions on surfaces, which are what make catalysts work, was only discovered many years later by Nobel Prize winner Prof. Gerhard Ertl, who transformed catalyst research from “black magic into an exact science”.
Despite the time it has taken to understand the technology, ammonia synthesis forms the basis for heterogeneous catalysis which is absolutely indispensable in modern chemical production. The lion’s share of the roughly 25,000 processes used to make bulk chemicals only work if the turbo is in the reactor.
Although chemical catalysts tend to be low-priced, BASF estimates that the worldwide market for catalysts in 2010 was in the region of €4 billion. The list of companies which reap the benefits from the chemical catalyst market includes BASF, Degussa Evonik, Süd-Chemie, Johnson Matthey, Heraeus and a number of Asian companies.
(ID:32307000)



:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/ea/43ead0a748c8f8659f0c7a73dcdc6977/0131324453v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/8e/848e1ad0b5c2943c6f27786f7676f7c6/0131301691v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/f5/97f5733274e1b148fd00e232e5d88cd0/0131301656v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/99/52/99524083a5317a2ca3de1b6f57394e73/0131300941v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/90/0990b4f93bc2629e6a76dd8d10c7ef9b/0131365319v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/18/42/1842c51b9071515986da6d691978da02/0131330223v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d1/2a/d12a316ecc0072ce9198954f46372971/0131327395v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/93/62/936234ea333c6a8bd476d40a4fbececf/0131365588v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8f/f4/8ff4c50de625c1ec2466280ff2de339a/0131364484v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/74/a1/74a1f89c92a80489f70daccbe7c26885/0131340042v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a7/44/a744d959fdf84c1bb5df81b75845c22a/0131301680v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/5f/98/5f98fbb2e7bf2/05-trm-filter-logo-with-slogan-en-alt.png)
:quality(80)/p7i.vogel.de/wcms/8c/6d/8c6d17e41698f8eca0342de145f8dff2/0127366713v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6e/db/6edb77bc47ad1638299792e211af183b/0124855705v1.jpeg)