Explosion protection Old Truth Stands no Longer: How dangerous is splash filling really?
One of the basic mantras when handling flammable liquids is to avoid free falling and splash filling at all costs. Dangerous electrostatic charges could otherwise cause an explosion with devastating effects. But how substantiated are these concerns? Recent experiments in Sweden and France came up with surprising results.
Related Vendors
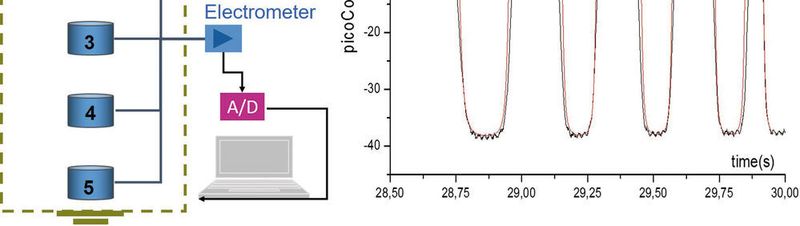
Filling tanks or containers with liquids is frequently done in the chemical industry. In many cases, the liquid is filled via a pipe ending at a distance above the bottom of the container and therefore may splash to the bottom or liquid surface. This process has often been blamed to generate dangerous discharges that may ignite dust or gases in the receiving tank.
Little has been known about the electrostatic charge that is generated through the splash filling process. Some experts claim that the Lenard effect, which states that electrostatic charges are generated when drops of a polar liquid are torn, could provide for an ignition source. Some scientists assume that falling droplets are charged by their contact with air molecules and thus create electrostatic hazards during splash filling.
To overcome this issue, first of all some theory is required: It is commonly assumed that electrostatic charging is caused by friction. This, nevertheless, is an over-simplification: In fact, charging is actually generated by separating materials which were in close contact before, if at least one of them is electrically insulating. As these charges occur at the material boundaries, so that the formation of interfaces is an absolute.
While these are natural for solid materials, liquids form them at their free surfaces. Only gases show no interfaces. Therefore, charge may occur at interfaces of solid/solid, solid/liquid and liquid/liquid but not between gases on the one hand and liquids or solids on the other.
Experience seems to contradict this fact as it is given proof that liquids will be electrostatically charged when being atomized, for example in air. But is there really a conflict? Not at all because when atomizing a liquid it will be disaggregated and at the interfaces resulting electrostatic charging comes into being, regardless of its conductivity and not by friction with the surrounding air.
(ID:44450237)



:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/8e/848e1ad0b5c2943c6f27786f7676f7c6/0131301691v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/f5/97f5733274e1b148fd00e232e5d88cd0/0131301656v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/99/52/99524083a5317a2ca3de1b6f57394e73/0131300941v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/55/c8/55c801c121267a30eedeb6afe339f233/0131300930v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/48/54/48540b44b56da638cbfafa7fb05dd4a2/0131245000v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b4/e5/b4e5eecb07d3491bde7bf6a22602ab77/0131085753v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/a6/43a69c0b9e26996ab4f3971f202afd82/0131055378v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a7/44/a744d959fdf84c1bb5df81b75845c22a/0131301680v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/79/7179c77f8bae9e48bf427708573ad6ac/0131057546v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3f/7f/3f7f7542cd5392be0f3074f074a75b36/0131055389v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/109600/109698/65.png)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/2000/2093/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/68/c8/68c815bc8fe81/prominent-logo-300x300.jpeg)
:quality(80)/images.vogel.de/vogelonline/bdb/1199400/1199486/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1199400/1199487/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1199400/1199488/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1199400/1199489/original.jpg)
:quality(80)/p7i.vogel.de/wcms/c1/ed/c1ed89188a8fdb351601e36204d4c38b/0125059797v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)