Drying and Cooling Fully Automatic Drying and Cooling with a new condensation drying system
Producing pharmaceuticals requires such a multitude and complexity of aspects to be considered that the actual drying process stage is often overlooked. This is despite the fact that high quality and reproducible drying can improve product quality while at the same time optimising the entire production process. This is what a major pharmaceutical company experienced when they implemented a condensation drying system.
Related Vendors
Takeda is a research-driven global enterprise mainly engaged in pharmaceuticals. Being Japan’s biggest pharmaceuticals manufacturer and among the globally leading companies in the field, Takeda is committed to improving the healthcare of patients all over the world through innovation in medicine.
The company is represented in some 70 countries with a focus on gastroenterology, oncology, cardiovascular and metabolic diseases, urology, gynaecology, immunological diseases, diseases of the central nervous system, general medicine and vaccines. The integration of Millennium Pharmaceuticals and Nycomed allowed Takeda to expand both geographically and in new therapeutic fields.
High International Standards
Since 2012 Takeda have managed their activities on the German market from their base in Berlin complemented by additional administrative functions in Constance. As part of their global production network, Takeda run production facilities at Oranienburg, Brandenburg, and at Singen. The latter site is specialised in lyophilised powders and semisolid dosage forms such as creams and balms. These substances are mostly used in oncology and gastroenterology.
“Takeda stand up for very high standards as regards quality”, says Thomas Griem, Takeda Engineering Project Manager. This quality mindedness was reflected at each single production stage, particularly when it came to optimising the drying process of the vials produced. Their previous drying technology no longer met Takeda’s requirements. By recommendation, Takeda learned of the Allgau based drying system manufacturer Harter. Upon a visit to the Harter site, Takeda was introduced to the Harter developed drying technology and agreed to have drying tests run in the pilot plant station.
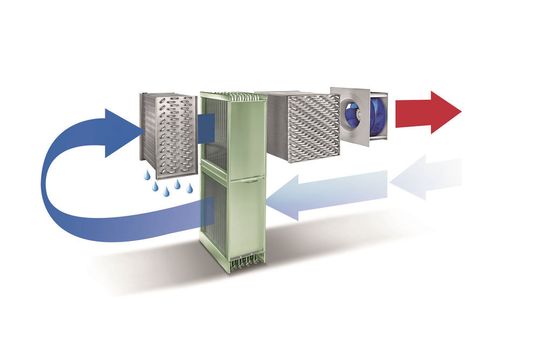
The Harter purpose developed drying method is the so-called “heat pump based condensation drying”. It combines seemingly conflicting features such as low temperatures and short drying times.
(ID:43972366)



:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/8e/848e1ad0b5c2943c6f27786f7676f7c6/0131301691v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/f5/97f5733274e1b148fd00e232e5d88cd0/0131301656v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/99/52/99524083a5317a2ca3de1b6f57394e73/0131300941v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/55/c8/55c801c121267a30eedeb6afe339f233/0131300930v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/48/54/48540b44b56da638cbfafa7fb05dd4a2/0131245000v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b4/e5/b4e5eecb07d3491bde7bf6a22602ab77/0131085753v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/a6/43a69c0b9e26996ab4f3971f202afd82/0131055378v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a7/44/a744d959fdf84c1bb5df81b75845c22a/0131301680v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/79/7179c77f8bae9e48bf427708573ad6ac/0131057546v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3f/7f/3f7f7542cd5392be0f3074f074a75b36/0131055389v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/5f/98/5f98fbb2e7bf2/05-trm-filter-logo-with-slogan-en-alt.png)
:quality(80)/images.vogel.de/vogelonline/bdb/1018800/1018896/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1018800/1018894/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1018800/1018897/original.jpg)
:quality(80)/images.vogel.de/vogelonline/bdb/1018800/1018898/original.jpg)
:quality(80)/p7i.vogel.de/wcms/aa/9f/aa9f87b6bd7ccdd61e3a0777ef37f701/0128191833v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3b/86/3b86c49f99e3638ed9f42c6cc7732950/0130491017v2.jpeg)