Related Vendors
The leap from the lab environment to industrial-scale production was the result of teamwork: Five partners from the scientific community and industry joined forces in the Dream Production project, which received government funding. Bayer Technology Services, Bayer Material Science, the power utility company RWE, the RWTH Aachen University and the CAT Catalytic Center, a research institution which is located on the RWTH campus and is jointly run by the university and Bayer, joined forces in the research consortium.
In the process, a pilot line was constructed at Bayer’s main site in Leverkusen in 2011. The CO2 for these trials, supplied from an RWE Niederaußem power station near Cologne, was reacted with propylene oxide to produce a high-viscosity, colorless mass, namely polyol.
:quality(80)/images.vogel.de/vogelonline/bdb/576800/576841/original.jpg)
Innovations
Signs of Life from the Lab: How Innovative is the Process Industry?
Kick-Off for a Dream
Now, following three years of pilot production, the time has come to scale up the process: A production line with a capacity of 5000 t/a will be built at an existing Bayer polyether plant in Dormagen. “The production volumes are relatively modest. The main emphasis is on further development of the technology,” explained Malsch, who is in charge of the Dream Production project.
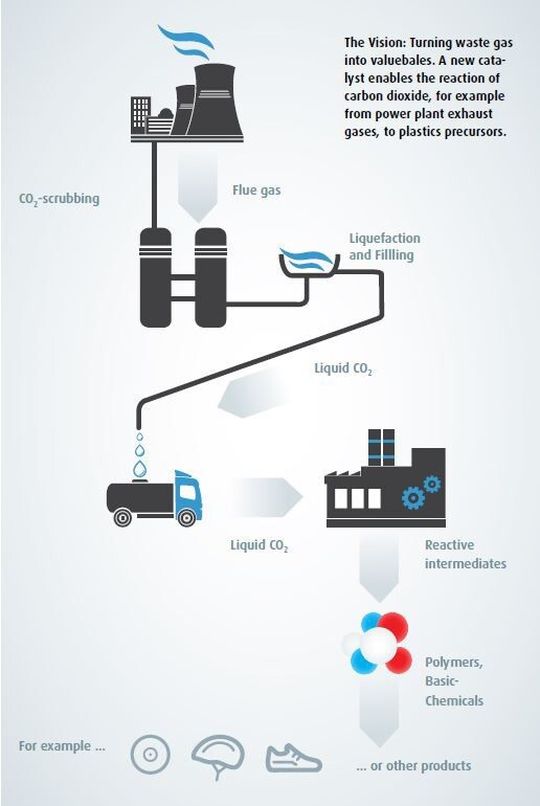
The fact that the plant in Dormagen is located at an integrated chemical site enables Bayer to exploit significant synergies. The CO2 needed for production is supplied as a waste product from a nearby Kemira ammonia plant. Starting in 2016, the plan is to begin using polyol produced on the line to make polyurethane foam for products such as heat insulation, shoes, auto parts, mattresses and furniture cushions.
A Better CO2–Balance Can Be Achieved
“Initially, production will be operating at a loss. We are investing up front, but over the medium term we expect to make money from the polyurethane when we ramp up production to the next level.” Bayer claims that the quality of the new material is at least as good as that of feedstock produced using conventional techniques, and there is an added sustainability dimension: By using a certain amount of CO2, production is no longer completely dependent on oil-based propylene oxide, thus reducing oil consumption. At any rate, the CO2 balance of the new process is better compared to the conventional production technique.
(ID:42706793)



:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/8e/848e1ad0b5c2943c6f27786f7676f7c6/0131301691v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/f5/97f5733274e1b148fd00e232e5d88cd0/0131301656v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/99/52/99524083a5317a2ca3de1b6f57394e73/0131300941v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/55/c8/55c801c121267a30eedeb6afe339f233/0131300930v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/48/54/48540b44b56da638cbfafa7fb05dd4a2/0131245000v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b4/e5/b4e5eecb07d3491bde7bf6a22602ab77/0131085753v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/a6/43a69c0b9e26996ab4f3971f202afd82/0131055378v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a7/44/a744d959fdf84c1bb5df81b75845c22a/0131301680v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/79/7179c77f8bae9e48bf427708573ad6ac/0131057546v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3f/7f/3f7f7542cd5392be0f3074f074a75b36/0131055389v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/61/f9/61f9439b7cbe4/logo-edl.png)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/103000/103097/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/68/c8/68c815bc8fe81/prominent-logo-300x300.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/b1/c8b1d42480b31c8048984d7349245a24/0128406704v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8c/6d/8c6d17e41698f8eca0342de145f8dff2/0127366713v2.jpeg)