Related Vendors
Toluene and Propyl Acetate Prone to Electrostatic Ignition
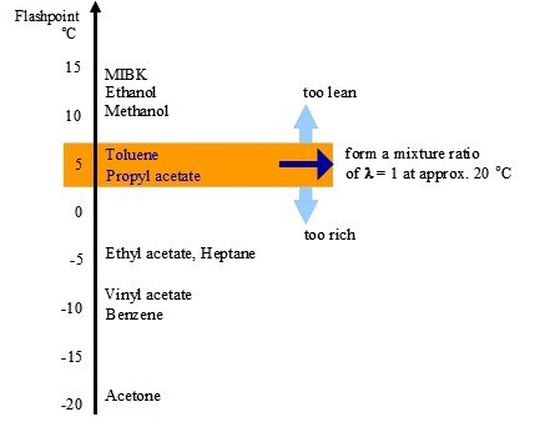
The mixing ratio created above a flammable liquid is determined largely by its flashpoint (FP). For example the mixtures of benzene are too rich (FP < -40 °C), on the other hand those of xylene FP = 28°C are too lean.
Particularly significant are the properties of toluene and propyl acetate. The flashpoint of both are at 5 °C, leading to a mixture ratio of aboutλ = 1 at room temperature. Thereby both liquids may be optimally ignited by electrostatic charges. However, propyl acetate shows a much better conductivity than toluene and, accordingly, is electrostatically charged much less.
Assuming that most refilling procedures take place at room temperature it becomes obvious that toluene is more often ignited electrostatically than other flammable liquids.
In the four following case histories, refilling procedures with toluene will be investigated at which fires occurred. All of them took place at room temperature. In view of these case histories it should be noted that the cause of an ignition can be proven only with more or less probability.
1. How to Transfer Toluene Into a Plastic Drum
Toluene from a distilling apparatus is being collected in an intermediate container and emptied into metal drums. Because of prevailing explosion danger in the area appropriate safety measures against electrostatic ignition dangers were taken:
- Filling hose and fill nozzle conductive and earthed
- Steel drums earthed
- Flooring dissipative
- Safety shoes dissipative
As all of a sudden no metal drum was available any more the worker took an empty plastic drum standing nearby, so that emptying of the intermediate container did not stagnate.
(ID:42843349)



:quality(80)/p7i.vogel.de/wcms/97/ff/97ffc5d8894eaaabac89fa86c88b5c04/0130558883v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/f4/54f4b4e2a86b203e0dfd84221602bdb5/0130367283v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/ae/54aece5dff672ce1651470eecd9f2589/0130138090v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d6/f7/d6f73325e96a269604cb98c8d10d25e1/0130703531v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/0c/9a/0c9ae4cc481eacac6b8857dfc2266aed/0130701785v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d6/c4/d6c445a05ff0384818acac8b3530e2d3/0130701377v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/5d/575d8ae4e7f56748c0aa48ca34735b5d/0130646451v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3b/86/3b86c49f99e3638ed9f42c6cc7732950/0130491017v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/78/8478fcee08360056b226cb8d5032d50c/0130489771v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ab/c2/abc21b4affa79498712fe95c16991828/0130489183v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/41/c9/41c96e0244221ad38daa73bdd8231477/0130477173v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/7d/0e/7d0e9e3fa6a5289338833948b8c8e6d0/0130702623v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/33/1c33cb645b2d07dacd829b45e08a932a/0130641492v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/98/989863664c7b65e87caf74a08239d487/0130167217v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/64/4e/644e22b71e8d35a63f051f781eb13886/0130163451v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/95/92/9592b332b377badeabde54aeba243ab5/0130644826v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8e/f9/8ef9bb7b46f133439b299c2ea2f4bac4/0130548981v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/0f/11/0f11c60729f25db0ca0d301d9d4ca0ee/0130485703v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/0c/a1/0ca135e0486c9af90a0552aea85de88c/0130439525v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b3/2f/b32fb9f5aa54e9cb6c92e4b6c8665263/0130163018v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/e3/09e378194b496921d2a267a6752322eb/0130333473v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/77/9d/779db435e7f9d79bafe16101008a9cfa/0130263548v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/109600/109698/65.png)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:quality(80)/p7i.vogel.de/wcms/bd/97/bd97a5da962cc860b64d427d2696881e/0125226771v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1e/f1/1ef1faa6b22814f36607c184a6b2d21c/0130164915v2.jpeg)