Related Vendors
The endpoint of the reaction between the acid components present in the sample and the alkaline solution (titrant) can be indicated by the color change of the indicator solution (such as phenolphthalein). Nowadays, electrochemical sensors have replaced the classical color indicators in titration analysis.
In fact, the electrochemical potential can be monitored conveniently by means of a pH glass electrode connected to automated titration instruments. Based on the potential measured in the sample, the addition of titrant is controlled. This means that larger or smaller portions (= increments) of alkaline titrant are added automatically by a motorized piston burette according to the signal change until the endpoint is reached.
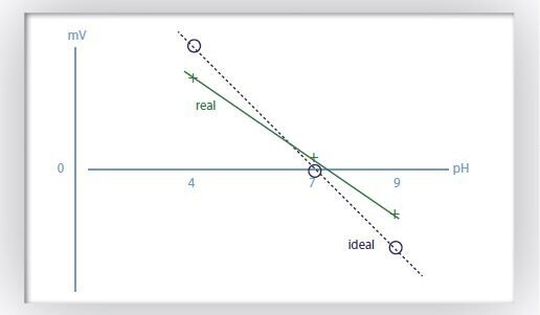
From the titrant consumption and its concentration the acid content can be calculated. As an example, the total acidity in water is determined by direct titration with diluted sodium hydroxide solution to pH 8.20. The total acidity of water is expressed in mmol/L or in mg/L calcium carbonate (CaCO3). Acidity of fruit juices is usually expressed in mg/L of a particular acid, e.g., citric acid.
How to Determine the Sugar Content
There are many different sugars, e.g. sucrose, malt sugar, glucose and HFCS. Strictly speaking, Brix only refers to sucrose content in aqueous solution. The unit Brix is defined as percentage by weight of sucrose in pure water solution. Therefore, the designation of Brix degrees is only valid for pure sucrose solutions in water. When determining the Brix degrees on malt sugar, glucose or honey or other sugars, the obtained results are not true Brix degree but related values only.
Furthermore, very accurate results can only be obtained by thermostating the sample to the required temperature (e.g., 20°C). This takes a long time with pycnometers or hydrometers. Digital density meters have a built-in Peltier thermostat which sets the temperature of the sample to ±0.02°C of the target temperature within less than a minute.
Raw refractive index and density values are converted into Brix degrees using official conversion tables issued by ICUMSA (International Commission for Uniform Methods of Sugar Analysis) and NBS (National Bureau of Standards), respectively As the reading depends on temperature a temperature compensation is needed to get accurate results. For manual instruments this implies adding a correction term to the reading. Digital handheld density and refractometers offer a built-in temperature correction, which obsoletes the manual error-prone compensation.
(ID:43164568)



:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/7e/c87e276b243339dbff4a8eca8f67a55e/0130871216v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/ff/97ffc5d8894eaaabac89fa86c88b5c04/0130558883v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/54/f4/54f4b4e2a86b203e0dfd84221602bdb5/0130367283v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e8/c4/e8c4c5839f7181878a3a76e8e463cf75/0130962757v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/58/e5/58e54f6f55b7318fd6afa201829f4b1e/0130957549v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/41/a7/41a7182a73ce7fe10ca37bb673609745/0130955359v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/74/247458cafd62b008794db34bda64df92/0130953253v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e0/2e/e02e124a066eb32e81e6ad35e6b32681/0130798212v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3b/86/3b86c49f99e3638ed9f42c6cc7732950/0130491017v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/78/8478fcee08360056b226cb8d5032d50c/0130489771v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ab/c2/abc21b4affa79498712fe95c16991828/0130489183v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/7d/0e/7d0e9e3fa6a5289338833948b8c8e6d0/0130702623v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d6/c4/d6c445a05ff0384818acac8b3530e2d3/0130701377v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/5d/575d8ae4e7f56748c0aa48ca34735b5d/0130646451v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/33/1c33cb645b2d07dacd829b45e08a932a/0130641492v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/98/989863664c7b65e87caf74a08239d487/0130167217v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/94/0e/940e978ce75be13842063b9c81d5f66e/0130871936v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bb/d8/bbd8135709bd2fae7fe813bc268feea0/0130871485v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/4b/d9/4bd9d4ef05d8bd7fdf78eca991ced8de/0130871466v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cf/cf/cfcff838134b67252b7f47d6cd8c84e4/0130871239v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/0c/a1/0ca135e0486c9af90a0552aea85de88c/0130439525v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/e3/09e378194b496921d2a267a6752322eb/0130333473v2.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/107800/107832/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/68/07/6807ad98568ce/logo-elementar-rgb.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/68/c8/68c815bc8fe81/prominent-logo-300x300.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/6f/276f0df09fd968e74c7c58d5915d07f1/0127144840v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c1/ed/c1ed89188a8fdb351601e36204d4c38b/0125059797v2.jpeg)