Microbial Testing Peace of Mind for Microbial Bioburden Testing
Merck has launched its latest innovation in pharmaceutical bioburden and water testing with its new Milliflex Oasis System.
Related Vendors
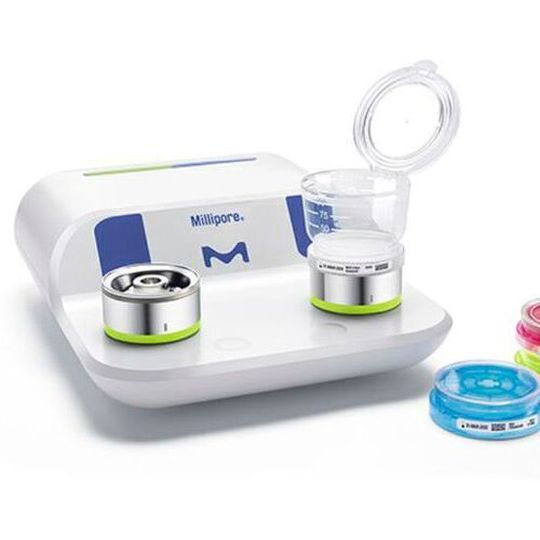
For water and bioburden testing, membrane filtration is the method of choice and accuracy is key to prevent wasting valuable time on false positive or false negative results. Merck has taken time to closely observe the complete membrane filtration workflow, to identify improvement possibilities and develop, hand-in-hand with users, a solution to simplify and safeguard the microbial testing process.
The Milliflex Oasis System, the company’s latest system has 96 new features to increase result reliability and traceability while streamlining the bioburden testing workflow.
According to the company, benefits of these upgrades include:
• Operator-independent handling steps, improved touch-free membrane transfer with colour-coded components for increased result reliability
• 50 per cent faster filtration time compared with previous product generation
• Scalable hardware for high throughput
• Reduced decontamination and maintenance time for optimised productivity
• 2-dimensional codes and labels on primary and secondary packaging, on each funnel and media plate, for traceability and regulation compliance
The company’s new system offers an all-in-one filtration solution with sterilised, ready-to-use filtration units and pharmacopeia-compliant media for comfort and peace of mind for water and bioburden testing.
(ID:46350267)



:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/91/4091929a36bbe16e0e0a5e58b3c46d64/0131194558v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/33/87/33873af45a622686767b0e597d41e7a3/0130872234v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/7e/c87e276b243339dbff4a8eca8f67a55e/0130871216v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1a/64/1a64a67b53537b893f5a6509dee97d87/0131192946v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/72/f3/72f3a2fed9f27d1b33a66f3021eb6ed2/0131192934v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6d/2c/6d2c5ab1d18334052a5f173a99758815/0131084924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/9a/6b9a2f9d820c458d6518f7d3c0cc990e/0131057985v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/48/54/48540b44b56da638cbfafa7fb05dd4a2/0131245000v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b4/e5/b4e5eecb07d3491bde7bf6a22602ab77/0131085753v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/a6/43a69c0b9e26996ab4f3971f202afd82/0131055378v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/79/7179c77f8bae9e48bf427708573ad6ac/0131057546v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3f/7f/3f7f7542cd5392be0f3074f074a75b36/0131055389v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/26/0e/260ee8cbbc76b6dcaa4d6221ecb44506/0131014543v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/94/0e/940e978ce75be13842063b9c81d5f66e/0130871936v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/61/f9/61f9439b7cbe4/logo-edl.png)
:fill(fff,0)/p7i.vogel.de/companies/5e/3c/5e3c3767b3d25/logo2.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:quality(80)/p7i.vogel.de/wcms/5c/27/5c27bddaef122b61c636e31d82a33bd9/0129017834v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ae/d2/aed2f3255c6e3cb5442310bda494a209/0127071524v2.jpeg)