UK: Growth Strategies Big Pharma Companies Conquer Orphan Drug Market Through M&As
To succeed in the lucrative orphan drug market, pharma companies require specialist capabilities. To do so, most big pharma companies have inherited specialty business units through mergers and acquisitions (M&As), says analytics company Global Data.
Related Vendors
London/UK — Global Data has identified the top 15 pharma players in 2025, according to forecast sales for their respective therapies, with orphan drug designations. The collective sales data from these therapies with orphan drug designations reveal that Roche has generated the highest sales and will maintain its leading position going forward. Maura Musciacco, Senior Director of Neurology and Ophthalmology at Global Data, explains that this could be largely attributed to its oncology portfolio, which has yielded and would continue to provide a significant proportion of the company’s sales.
The vast majority of Roche’s oncology drugs with an orphan drug designation are expected to be blockbusters by 2025, including Perjeta (pertuzumab), Tecentriq (atezolizumab), Herceptin (trastuzumab), Avastin (bevacizumab), Rituxan (rituximab), Venclexta (venetoclax), Kadcyla (ado-trastuzumab emtansine), Alecensa (alectinib) and Gazyva (obinutuzumab). Musciacco notes that none of these drugs were originally developed in-house, but were gained through successful M&As, as exemplified by the acquisition of one of the leading biotech players, Genentech.
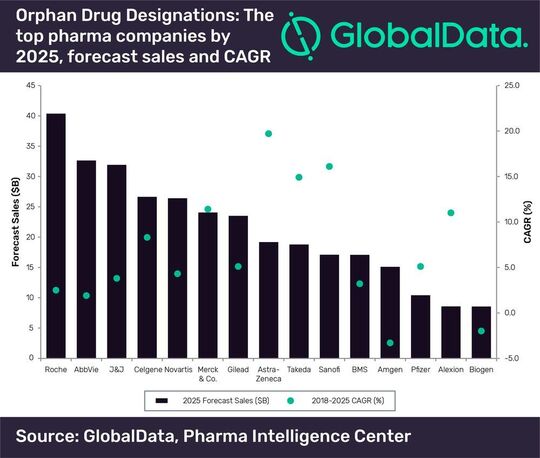
The only players that will have double-digit sales growth will be Merck (11.4 %), Astra Zeneca (19.7), Takeda (14.9 %), and Sanofi (16.1 %). According to Musciacco, Astra Zeneca is expected to have the strongest CAGR for its therapies with orphan drug designations over 2018–2025. This can also be attributed to its oncology offering, specifically the oncology drugs that were originally developed by other specialist players acquired by the company, such as Imfinzi (durvalumab), which was developed by MedImmune; Lynparza (olaparib), which was developed by Kudos Pharmaceuticals; and Calquence (acalabrutinib), which was developed by Acerta Pharma.
Takeda is also expected to see strong sales growth from its therapies with orphan drug designations. Of these, the ones with the strongest CAGRs were inherited from its M&As with Shire, Ariad Pharmaceuticals and Millennium Pharmaceuticals.
At the other end of the spectrum, the only players that are expected to have negative CAGRs from their therapies with orphan drug designations are Amgen (-3.3 %) and Biogen (-2 %). In both cases, this could be attributed to their mature brands, declining sales due to cannibalization from generics or biosimilars, and ultimately the failure to replace their old flagship brands with novel pipeline drugs, says Musciacco. In a defensive strategy, Amgen and Biogen were expected to turn to M&As in the near future as a means to maintain their respective presence in the orphan drugs sector and rejuvenate their pipelines by inheriting the expertise from niche specialist players, he added.
:quality(80)/images.vogel.de/vogelonline/bdb/1608000/1608096/original.jpg)
Global Pharma Sector
Growth Snapshot of Top 20 Pharma Companies
(ID:46111243)



:quality(80)/p7i.vogel.de/wcms/f1/41/f141fc30eddadc5a340d28fd8980aa27/0131438828v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/22/a4/22a4a5a34b3157c9ef8b6fdc5a2b11ec/0131429879v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/57/2757786ef0ad2d230e7776df6079b28b/0131301702v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/59/6c/596cdde662a90ad3295fb67ad7f74737/0131244347v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b2/f9/b2f9f1b88b4ee749df0ea3af3a7da90d/0131440719v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/43/ea/43ead0a748c8f8659f0c7a73dcdc6977/0131324453v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/84/8e/848e1ad0b5c2943c6f27786f7676f7c6/0131301691v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/f5/97f5733274e1b148fd00e232e5d88cd0/0131301656v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f5/ea/f5eae046dd5abe4a43b65e4aee1d5784/0131429900v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/85/7c/857c62a32c14d9cc02310ffe5a6b1f17/0131301667v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c6/20/c620db13d7331aa76791b0742c47a9b3/0131248252v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/45/6a45ee92ec1a9e84cffe55964a693b74/0131139927v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/09/90/0990b4f93bc2629e6a76dd8d10c7ef9b/0131365319v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/18/42/1842c51b9071515986da6d691978da02/0131330223v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d1/2a/d12a316ecc0072ce9198954f46372971/0131327395v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ff/37/ff3789529a54269859a36f49352f9ac8/0131245949v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/e9/64/e964996410967c124c2cf01a8623068e/0130285341v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/1c/39/1c39433f4bb4de2e5e6c5311c3236d09/0130110254v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/e1/bee13b8d51a419b9b8f0685e2b9d47bf/0128917673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/7a/517a4e83c5827663b4839f5ddb98f434/0128872953v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b7/68/b76861f18db2d568329cdd966cf23e90/0131083977v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fa/0e/fa0efd09be99b54318c553d9e8dcb52d/0130922390v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/ef/60efa975d1dd123c1afbfb6ccc060461/0130263955v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/62/0762f20b1821ee2724c97989374d6397/0130257651v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/31/7931cc94924b4c2ebc7087e9cec2f801/0130490720v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/cc/6bcc3d26e1c7c74d90a10bc3ca296ae1/0128362351v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/24/c8/24c84ab6b1bcd24468a87820a07a85aa/0128194707v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/1d/3e/1d3e2788eb37c1e974f36337c8d33cb5/0128191010v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/45/f2/45f2f4b9e76e820ad1a12c4342920260/0131439171v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/66/c9/66c916cef9c4e9f170ca46972d8c35b3/0131429703v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/93/62/936234ea333c6a8bd476d40a4fbececf/0131365588v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8f/f4/8ff4c50de625c1ec2466280ff2de339a/0131364484v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/29/f8298429c1638b949a6f76346f6709c7/0118701710v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/07/24/07242664ab2b1c7841c9d9d0a127670c/0116045959.jpeg)
:quality(80)/p7i.vogel.de/wcms/c9/79/c979a20b32395ddfa93fe7ead90578a0/0108386061.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/5c/9e5c92d942ed046a27562d6e3d730c92/0103483548.jpeg)
:quality(80)/p7i.vogel.de/wcms/57/da/57da73af9c35d633196afd46348182b2/0130820106v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/36/20366ffa57a841882fd165e4dc885c09/0130491929v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d0/f7/d0f7d560dfce37c8371442d5b6120e3c/0130182478v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/7c/77/7c77e24fdcc9e1511e8e711eff161e28/0130175489v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/a8/4e/a84e8039a90a5cf4751d01ebcf6ba1a9/0127510172v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/17/c7/17c703445f134eb3d7ecc7918dda2762/0124596096v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/2c/2a2cffc07f51019065387cd63241b5ce/0119463370v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/7e/b17ea8c62ccafad1d1fb072d6199bbd6/0118578446.jpeg)
:quality(80)/p7i.vogel.de/wcms/6a/7e/6a7e738aac81597b04bae84add8a9077/0131141034v2.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/5e/a2/5ea2b5ca18eac/schubert-cp-gross-rgb--003-.jpg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/101800/101879/65.jpg)
:fill(fff,0)/p7i.vogel.de/companies/5f/98/5f98fbb2e7bf2/05-trm-filter-logo-with-slogan-en-alt.png)
:quality(80)/p7i.vogel.de/wcms/3d/43/3d437a40d6f10dcfd3017ba721feb16a/0127144444v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/41/d5/41d5d18ea6b865adab1d6cb18d19cbdc/0128416809v1.jpeg)